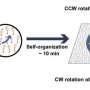
Research has unveiled that chiral myosin can steer actin into stable rotating rings without relying on a template. This finding enhances our understanding of how living cells achieve organization through dynamic molecular interactions. The study illustrates that cellular order is not the result of fixed blueprints but emerges from the continuous movements and rearrangements of molecules.
Living cells exhibit a remarkable level of organization, which has intrigued scientists for decades. The latest research highlights the significance of chiral myosin, a type of motor protein, in facilitating the formation of actin structures. Actin, a vital component of the cell’s cytoskeleton, plays a crucial role in maintaining cell shape and enabling movement.
Understanding the Mechanism of Asymmetry
One of the most notable phenomena observed in living organisms is left-right asymmetry, a feature that is evident in many biological structures. This study sheds light on the underlying mechanisms that contribute to this asymmetry. According to the research team, the interactions between chiral myosin and actin lead to the spontaneous formation of stable rotating rings.
Through a series of experiments, the researchers demonstrated that the chiral nature of myosin directs the arrangement of actin filaments into these effective circular formations. This process does not require a predetermined template, suggesting that cellular organization can arise from inherent molecular properties rather than rigid structural guidelines.
Implications for Cellular Biology and Beyond
The implications of this research extend beyond basic cellular biology. Understanding how chiral myosin influences actin dynamics could have significant applications in various fields, including bioengineering and medicine. By harnessing these insights, scientists might develop new strategies for influencing cell behavior, which could be pivotal in areas such as tissue regeneration and cancer treatment.
The findings contribute to a growing body of literature that emphasizes the importance of molecular interactions in biological systems. The research was published in a reputable scientific journal, ensuring that it reaches the broader scientific community. As researchers continue to explore the complexities of cellular organization, studies like this one provide critical insights that could lead to innovative solutions to pressing biological challenges.
Overall, this study not only enhances our understanding of cellular mechanics but also opens the door to future research that could explore the potential applications of these findings in health and technology.