A new and highly potent weight loss drug called retatrutide has emerged in the market, raising significant health concerns as it is not yet approved for public use in any country. Often referred to as the “Godzilla of weight loss injections,” retatrutide operates similarly to existing medications like Wegovy and Mounjaro, which mimic gut hormones to suppress appetite. Early-stage trials suggest that retatrutide could lead to greater weight loss compared to these approved options, but without larger trials to confirm safety, its use poses serious risks.
Layla Moran, the chair of Parliament’s Health and Social Care Select Committee, has voiced alarm over the unregulated trade of retatrutide, stating that the lack of medical approval represents a “serious threat to public health.” In response, she has urged the Medicines and Healthcare Products Regulatory Agency (MHRA) to take action against the illegal trade of these weight loss injections.
Understanding Retatrutide’s Mechanism and Differences
Retatrutide distinguishes itself from other approved weight loss medications through its potency. While Wegovy, which contains semaglutide, typically results in an average weight loss of about 15 percent after one year, and Mounjaro, containing tirzepatide, can lead to a 21 percent reduction, retatrutide has shown potential for up to 24 percent weight loss after one year in smaller Phase 2 trials. Notably, the weight loss continued beyond this point, suggesting that final results could be even more significant.
The mechanism of retatrutide involves mimicking three hormones: GLP-1, GIP, and glucagon, which has led to its nickname of “triple G.” This complex interaction may account for its increased effectiveness, though the lack of larger trial data raises questions about long-term safety.
Risks of Unapproved Use
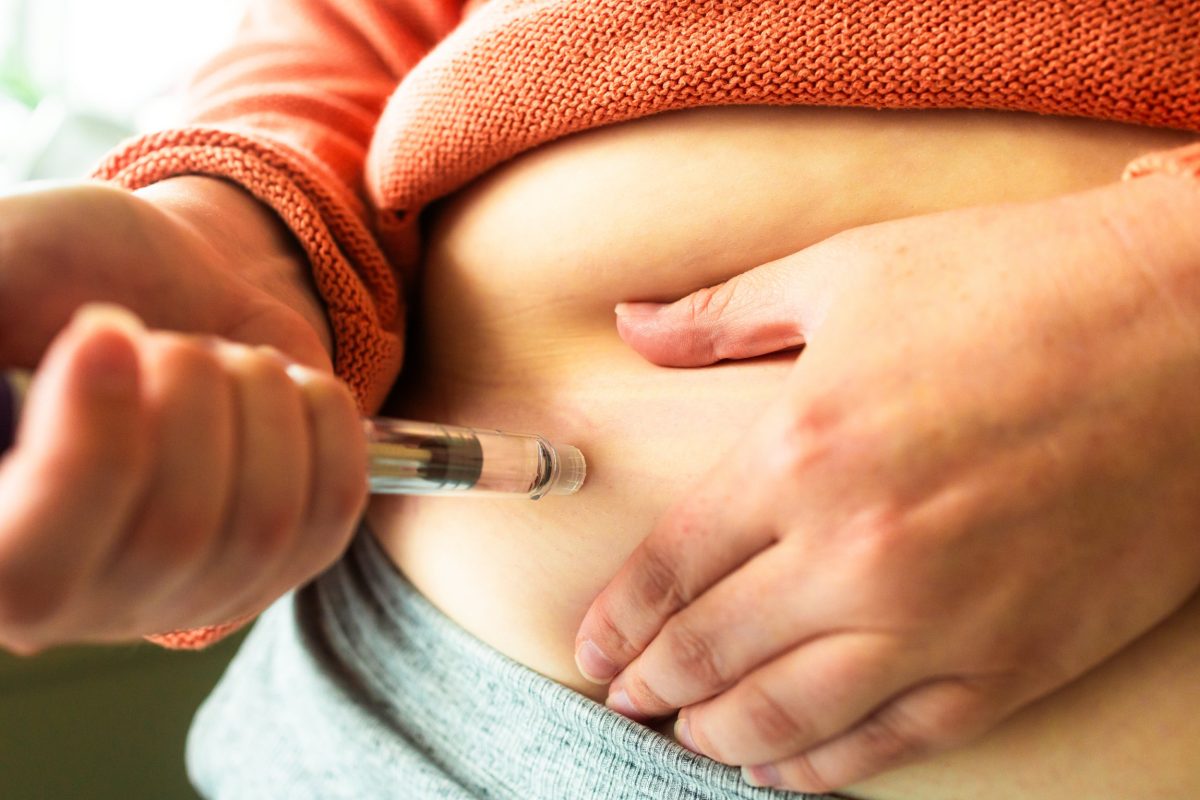
Despite the promising early results, retatrutide is not legally available for human use without authorization from its patent holder, Eli Lilly. The pharmaceutical company has reiterated that anyone marketing retatrutide for human consumption is violating the law. The chemical formula for retatrutide is publicly accessible, allowing small manufacturers, particularly in China, to create and sell the drug online, often under the guise of “research purposes only.”
These online sellers frequently offer the drug in powder form, requiring buyers to mix it with sterile water, or as pre-mixed vials. The appeal of retatrutide extends beyond weight loss, with some individuals using it to enhance muscle gain as well.
The risks associated with purchasing and using unapproved drugs are significant. Approved weight loss injections in the UK, when used under medical supervision, are generally considered safe. However, individuals who self-medicate with unregulated products face an increased likelihood of severe side effects, including nausea, vomiting, and diarrhea. More serious complications have also been reported, such as pancreatitis and, in rare cases, fatalities.
Moran highlighted these dangers in a letter to the MHRA, noting that the potential for serious side effects is heightened when using untested and unlicensed drugs. Kiran Jones, a pharmacist at Oxford Online Pharmacy, emphasized that without regulatory oversight, there is no assurance of product quality or safety.
Challenges in Regulation and Future Availability
Efforts to regulate the online sale of retatrutide have been met with challenges. Following a Channel 4 investigation, platforms like TikTok and Meta have removed advertisements promoting the drug, but links to sellers remain readily accessible. Eli Lilly has indicated it is pursuing action against two online sellers, although specific details have not been disclosed.
The final Phase 3 trials for retatrutide are anticipated to conclude in May 2024. If these trials confirm the drug’s safety and effectiveness, it could receive approval in the UK, potentially making it available by 2027. As the situation develops, the MHRA is expected to continue monitoring the unregulated market closely.