The National Health Service (NHS) has approved a revolutionary treatment, obe-cel, which has the potential to cure leukaemia. This innovative therapy, developed by Autolus, a spin-out company from University College London, works by genetically modifying patients’ cells to enhance the immune system’s ability to identify and combat cancer. Unlike traditional treatments, obe-cel requires only a single administration, consisting of two intravenous doses given ten days apart.
The National Institute for Health and Care Excellence (NICE) has recommended the use of obe-cel, also known as Aucatzyl, for adults over the age of 26 in England. This approval could provide hope for more than 150 patients within the next three years, significantly impacting those with leukaemia who have either relapsed after previous therapies or have not responded to initial treatments.
Clinical trials have demonstrated promising results, with a study involving 94 participants revealing that 77 percent achieved remission following treatment. Notably, over half of these patients remained cancer-free for more than three and a half years after receiving obe-cel.
Patient Experiences and Expert Insights
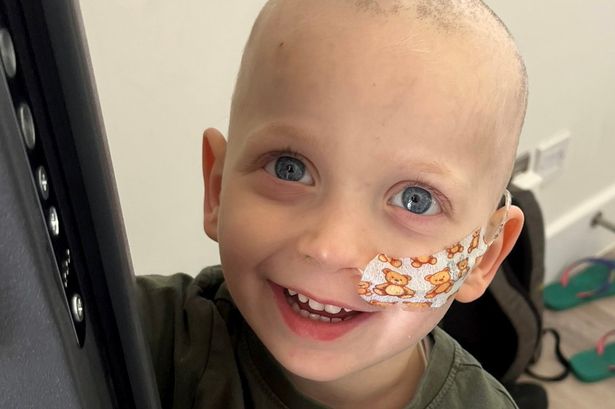
Among those who have benefited from this cutting-edge therapy is Harry Brown, a 19-year-old student from Harrogate. Having participated in a trial last year, he expressed his gratitude, stating, “I feel so lucky to have had access to such a wondrous treatment. Not only did it work better than my doctors thought, it did so without many of the horrible side effects you can get from other treatments.”
Dr. Claire Roddie, an expert at UCL, expressed her enthusiasm regarding NICE’s decision. She stated, “I’m delighted to hear of NICE’s decision. Many more patients now stand to benefit.” This sentiment was echoed by Leukaemia UK, which described the approval as “a significant step forward” in the fight against leukaemia.
The introduction of obe-cel adds to the limited options currently available for adult leukaemia patients, complementing a similar treatment that exists for those aged 25 and under. This advancement is not just a milestone in cancer therapy but represents a beacon of hope for patients and families grappling with the challenges of leukaemia.
As the NHS begins to roll out this treatment, the medical community remains optimistic about its potential to transform the lives of many, paving the way for further innovations in cancer care.